Our Health Library information does not replace the advice of a doctor. Please be advised that this information is made available to assist our patients to learn more about their health. Our providers may not see and/or treat all topics found herein. This information is produced and provided by the National Cancer Institute (NCI). The information in this topic may have changed since it was written. For the most current information, contact the National Cancer Institute via the Internet web site at http://cancer.gov or call 1-800-4-CANCER. Rectal cancer is a type of cancer that forms in the tissues of the rectum. The rectum is part of the body's digestive system. The digestive system takes in nutrients (vitamins, minerals, carbohydrates, fats, proteins, and water) from foods and helps pass waste material out of the body. The digestive system is made up of the esophagus, stomach, and the small and large intestines. The colon (large bowel) is the main part of the large intestine and is about 5 feet long. Together, the rectum and anal canal make up the last part of the large intestine and are 6 to 8 inches long. The anal canal ends at the anus (the opening of the large intestine to the outside of the body). Health history affects the risk of developing rectal cancer. Colorectal cancer is caused by certain changes to the way colorectal cells function, especially how they grow and divide into new cells. There are many risk factors for colorectal cancer, but many do not directly cause cancer. Instead, they increase the chance of DNA damage in cells that may lead to colorectal cancer. To learn more about how cancer develops, see What Is Cancer? A risk factor is anything that increases the chance of getting a disease. Some risk factors for colorectal cancer, like smoking, can be changed. However, risk factors also include things you cannot change, like your genetics, getting older, and your family history. Learning about risk factors for colorectal cancer can help you make changes that might lower your risk of getting it. Risk factors for colorectal cancer include: Older age is a main risk factor for most cancers. The chance of getting cancer increases as you get older. Having one or more of these risk factors does not mean that you will get colorectal cancer. Many people with risk factors never develop colorectal cancer, while others with no known risk factors do. Talk with your doctor if you think you might be at increased risk. Signs of rectal cancer include blood in the stool or a change in bowel habits. These and other signs and symptoms may be caused by rectal cancer or by other conditions. Check with your doctor if you have: Tests that examine the rectum and colon are used to diagnose rectal cancer. In addition to asking about your personal and family health history and doing a physical exam, your doctor may perform the following tests and procedures: After rectal cancer has been diagnosed, imaging tests are done to find out if cancer cells have spread within the rectum or to other parts of the body. The process used to find out whether cancer has spread within the rectum or to other parts of the body is called staging. The information gathered from the staging process determines the stage of the disease. It is important to know the stage in order to plan treatment. The following tests and procedures may be used in the staging process: Some people decide to get a second opinion. You may want to get a second opinion to confirm your rectal cancer diagnosis and treatment plan. If you seek a second opinion, you will need to get medical test results and reports from the first doctor to share with the second doctor. The second doctor will review the pathology report, slides, and scans. They may agree with the first doctor, suggest changes or another treatment approach, or provide more information about your cancer. Learn more about choosing a doctor and getting a second opinion at Finding Cancer Care. You can contact NCI's Cancer Information Service via chat, email, or phone (both in English and Spanish) for help finding a doctor, hospital, or getting a second opinion. For questions you might want to ask at your appointments, visit Questions to Ask Your Doctor About Cancer. Certain factors affect prognosis (chance of recovery) and treatment options. The prognosis and treatment options depend on: Cancer stage describes the extent of cancer in the body, such as the size of the tumor, whether it has spread, and how far it has spread from where it first formed. There are several staging systems for cancer that describe the extent of the cancer. Rectal cancer staging usually uses the TNM staging system. The cancer may be described by this staging system in your pathology report. Based on the TNM results, a stage (I, II, III, or IV, also written as 1, 2, 3, or 4) is assigned to your cancer. When talking to you about your diagnosis, your doctor may describe the cancer as one of these stages. Learn about tests to stage rectal cancer. Learn more about Cancer Staging. The following stages are used for rectal cancer: Stage 0 (carcinoma in situ) In stage 0 rectal cancer, abnormal cells are found in the mucosa (innermost layer) of the rectum wall. These abnormal cells may become cancer and spread into nearby normal tissue. Stage 0 is also called carcinoma in situ. Stage I (also called stage 1) rectal cancer In stage I rectal cancer, cancer has formed in the mucosa (innermost layer) of the rectum wall and has spread to the submucosa (layer of tissue next to the mucosa) or to the muscle layer of the rectum wall. Stage II (also called stage 2) rectal cancer Stage II rectal cancer is divided into stages IIA, IIB, and IIC. Stage III (also called stage 3) rectal cancer Stage III rectal cancer is divided into stages IIIA, IIIB, and IIIC. In stage IIIA, cancer has spread: In stage IIIB, cancer has spread: In stage IIIC, cancer has spread: Stage IV (also called stage 4) rectal cancer Stage IV rectal cancer is divided into stages IVA, IVB, and IVC. Stage IV rectal cancer is also called metastatic rectal cancer. Metastatic cancer happens when cancer cells travel through the lymphatic system or blood and form tumors in other parts of the body. The metastatic tumor is the same type of cancer as the primary tumor. For example, if rectal cancer spreads to the liver, the cancer cells in the liver are actually rectal cancer cells. The disease is called metastatic rectal cancer, not liver cancer. Learn more in Metastatic Cancer: When Cancer Spreads. Rectal cancer can recur (come back) after it has been treated. Recurrent rectal cancer is cancer that has come back after it has been treated. If rectal cancer comes back, it may come back in the rectum or in other parts of the body, such as the colon, pelvis, liver, or lungs. Tests will be done to help determine where the cancer has returned. The type of treatment for recurrent rectal cancer will depend on where it has come back. Learn more in Recurrent Cancer: When Cancer Comes Back. Information to help you cope and talk with your health care team can be found in the booklet When Cancer Returns. There are different types of treatment for people with rectal cancer. Different types of treatments are available for rectal cancer. You and your cancer care team will work together to decide your treatment plan, which may include more than one type of treatment. Many factors will be considered, such as the stage of the cancer, your overall health, and your preferences. Your plan will include information about your cancer, the goals of treatment, your treatment options and the possible side effects, and the expected length of treatment. Talking with your cancer care team before treatment begins about what to expect will be helpful. You'll want to learn what you need to do before treatment begins, how you'll feel while going through it, and what kind of help you will need. To learn more, visit Questions to Ask Your Doctor About Treatment. The following types of treatment are used: Surgery Surgery is the most common treatment for all stages of rectal cancer. The cancer is removed using one of the following types of surgery: After the cancer is removed, the surgeon will either: Radiation therapy and/or chemotherapy may be given before surgery to shrink the tumor, make it easier to remove the cancer, and help with bowel control after surgery. Treatment given before surgery is called neoadjuvant therapy. After all the cancer that can be seen at the time of the surgery is removed, some patients may be given radiation therapy and/or chemotherapy after surgery to kill any cancer cells that are left. Treatment given after the surgery, to lower the risk that the cancer will come back, is called adjuvant therapy. If the cancer has spread to the liver and cannot be removed by surgery, a total hepatectomy and liver transplant after chemotherapy may be done. Total hepatectomy and liver transplant is the removal of the entire liver by surgery, followed by a transplant of a healthy liver from a donor. Radiation therapy Radiation therapy uses high-energy x-rays or other types of radiation to kill cancer cells or keep them from growing. Rectal cancer is sometimes treated with external radiation therapy. This type of radiation therapy uses a machine outside the body to send radiation toward the area of the body with cancer. Short-course preoperative radiation therapy is used in some types of rectal cancer. This type of external radiation therapy uses fewer and lower doses of radiation than standard treatment, followed by surgery several days after the last dose. Learn more about External Beam Radiation Therapy for Cancer and Radiation Therapy Side Effects. Chemotherapy Chemotherapy (also called chemo) uses drugs to stop the growth of cancer cells, either by killing the cells or by stopping them from dividing. Systemic chemotherapy is when chemotherapy drugs are taken by mouth or injected into a vein or muscle. When given this way, the drugs enter the bloodstream and can reach cancer cells throughout the body. Systemic chemotherapy used to treat rectal cancer includes: Combinations of these drugs may be used. Other chemotherapy drugs not listed here may also be used. Chemotherapy may also be combined with other kinds of drugs. For example, it might be combined with the targeted therapy drug bevacizumab, cetuximab, or panitumumab. Regional chemotherapy for rectal cancer is when drugs are placed directly into the hepatic artery (the main artery that supplies blood to the liver) in a procedure called chemoembolization. Chemoembolization of the hepatic artery may be used to treat cancer that has spread to the liver. This is done by blocking the hepatic artery and injecting anticancer drugs between the blockage and the liver. The liver's arteries then carry the drugs into the liver. Only a small amount of the drug reaches other parts of the body. The blockage may be temporary or permanent, depending on what is used to block the artery. The liver continues to receive some blood from the hepatic portal vein, which carries blood from the stomach and intestine. The way the chemotherapy is given depends on the type and stage of the cancer being treated. Learn more about how chemotherapy works, how it is given, common side effects, and more at Chemotherapy to Treat Cancer and Chemotherapy and You: Support for People With Cancer. Chemoradiation therapy Chemoradiation therapy combines chemotherapy and radiation therapy to increase the effects of both. Active surveillance Active surveillance is closely following a patient's condition without giving any treatment unless there are changes in test results. It is used to find early signs that the condition is getting worse. In active surveillance, patients are given certain exams and tests to check if the cancer is growing. When the cancer begins to grow, treatment is given to cure the cancer. Tests include: Targeted therapy Targeted therapy uses drugs or other substances to identify and attack specific cancer cells. Your doctor may suggest biomarker tests to help predict your response to certain targeted therapy drugs. Learn more about Biomarker Testing for Cancer Treatment. Targeted therapies used to treat rectal cancer include: Learn more about Targeted Therapy to Treat Cancer. Immunotherapy Immunotherapy helps a person's immune system fight cancer. Your doctor may suggest biomarker tests to help predict your response to certain immunotherapy drugs. Learn more about Biomarker Testing for Cancer Treatment. Immunotherapy drugs used to treat rectal cancer include: Learn more about Immunotherapy to Treat Cancer. New types of treatment are being tested in clinical trials. For some people, joining a clinical trial may be an option. There are different types of clinical trials for people with cancer. For example, a treatment trial tests new treatments or new ways of using current treatments. Supportive care and palliative care trials look at ways to improve quality of life, especially for those who have side effects from cancer and its treatment. You can use the clinical trial search to find NCI-supported cancer clinical trials accepting participants. The search allows you to filter trials based on the type of cancer, your age, and where the trials are being done. Clinical trials supported by other organizations can be found on the ClinicalTrials.gov website. Learn more about clinical trials, including how to find and join one, at Clinical Trials Information for Patients and Caregivers. Treatment for rectal cancer may cause side effects. For information about side effects caused by treatment for cancer, visit our Side Effects page. Follow-up care may be needed. As you go through treatment, you will have follow-up tests or check-ups. Some tests that were done to diagnose or stage the cancer may be repeated to see how well the treatment is working. Decisions about whether to continue, change, or stop treatment may be based on the results of these tests. Some of the tests will continue to be done from time to time after treatment has ended. The results of these tests can show if your condition has changed or if the cancer has recurred (come back). After treatment for rectal cancer, a blood test to measure amounts of carcinoembryonic antigen (a substance in the blood that may be increased when cancer is present) may be done to see if the cancer has come back. Treatment of stage 0 may include the following types of surgery: Learn more about these treatments in the Treatment Option Overview. Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available. Treatment of stage I rectal cancer may include: Learn more about these treatments in the Treatment Option Overview. Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available. Treatment of stage II and stage III rectal cancer may include: Learn more about these treatments in the Treatment Option Overview. Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available. Treatment of stage IV and recurrent rectal cancer may include: Treatment of rectal cancer that has spread to other organs depends on where the cancer has spread. Learn more about these treatments in the Treatment Option Overview. Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available. For more information from the National Cancer Institute about rectal cancer, visit: For general cancer information and other resources from the National Cancer Institute, visit: About PDQ Physician Data Query (PDQ) is the National Cancer Institute's (NCI's) comprehensive cancer information database. The PDQ database contains summaries of the latest published information on cancer prevention, detection, genetics, treatment, supportive care, and complementary and alternative medicine. Most summaries come in two versions. The health professional versions have detailed information written in technical language. The patient versions are written in easy-to-understand, nontechnical language. Both versions have cancer information that is accurate and up to date and most versions are also available in Spanish. PDQ is a service of the NCI. The NCI is part of the National Institutes of Health (NIH). NIH is the federal government's center of biomedical research. The PDQ summaries are based on an independent review of the medical literature. They are not policy statements of the NCI or the NIH. Purpose of This Summary This PDQ cancer information summary has current information about the treatment of rectal cancer. It is meant to inform and help patients, families, and caregivers. It does not give formal guidelines or recommendations for making decisions about health care. Reviewers and Updates Editorial Boards write the PDQ cancer information summaries and keep them up to date. These Boards are made up of experts in cancer treatment and other specialties related to cancer. The summaries are reviewed regularly and changes are made when there is new information. The date on each summary ("Updated") is the date of the most recent change. The information in this patient summary was taken from the health professional version, which is reviewed regularly and updated as needed, by the PDQ Adult Treatment Editorial Board. Clinical Trial Information A clinical trial is a study to answer a scientific question, such as whether one treatment is better than another. Trials are based on past studies and what has been learned in the laboratory. Each trial answers certain scientific questions in order to find new and better ways to help cancer patients. During treatment clinical trials, information is collected about the effects of a new treatment and how well it works. If a clinical trial shows that a new treatment is better than one currently being used, the new treatment may become "standard." Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment. Clinical trials can be found online at NCI's website. For more information, call the Cancer Information Service (CIS), NCI's contact center, at 1-800-4-CANCER (1-800-422-6237). Permission to Use This Summary PDQ is a registered trademark. The content of PDQ documents can be used freely as text. It cannot be identified as an NCI PDQ cancer information summary unless the whole summary is shown and it is updated regularly. However, a user would be allowed to write a sentence such as "NCI's PDQ cancer information summary about breast cancer prevention states the risks in the following way: [include excerpt from the summary]." The best way to cite this PDQ summary is: PDQ® Adult Treatment Editorial Board. PDQ Rectal Cancer Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/colorectal/patient/rectal-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389378] Images in this summary are used with permission of the author(s), artist, and/or publisher for use in the PDQ summaries only. If you want to use an image from a PDQ summary and you are not using the whole summary, you must get permission from the owner. It cannot be given by the National Cancer Institute. Information about using the images in this summary, along with many other images related to cancer can be found in Visuals Online. Visuals Online is a collection of more than 3,000 scientific images. Disclaimer The information in these summaries should not be used to make decisions about insurance reimbursement. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page. Contact Us More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website's E-mail Us. Last Revised: 2024-09-13 If you want to know more about cancer and how it is treated, or if you wish to know about clinical trials for your type of cancer, you can call the NCI's Cancer Information Service at 1-800-422-6237, toll free. A trained information specialist can talk with you and answer your questions. This information does not replace the advice of a doctor. Ignite Healthwise, LLC disclaims any warranty or liability for your use of this information. Your use of this information means that you agree to the Terms of Use and Privacy Policy. Learn how we develop our content. Healthwise, Healthwise for every health decision, and the Healthwise logo are trademarks of Ignite Healthwise, LLC.Topic Contents
Rectal Cancer Treatment (PDQ®): Treatment - Patient Information [NCI]
General Information About Rectal Cancer
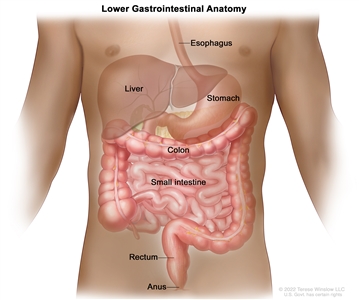
Anatomy of the lower gastrointestinal (digestive) system showing the colon, rectum, and anus. Other organs that make up the digestive system are also shown.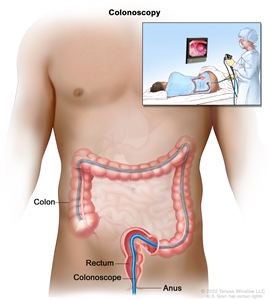
Colonoscopy. A thin, lighted tube is inserted through the anus and rectum and into the colon to look for abnormal areas.Stages of Rectal Cancer
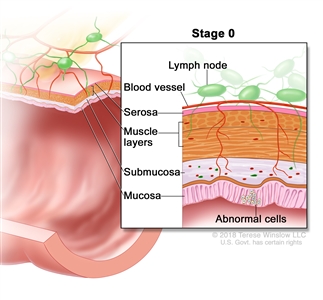
Stage 0 (rectal carcinoma in situ). Abnormal cells are shown in the mucosa of the rectum wall.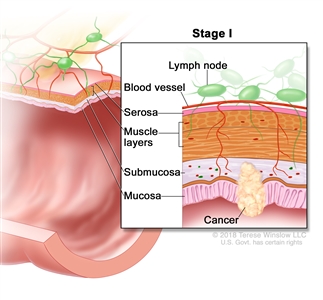
Stage I rectal cancer. Cancer has spread from the mucosa of the rectum wall to the submucosa or to the muscle layer.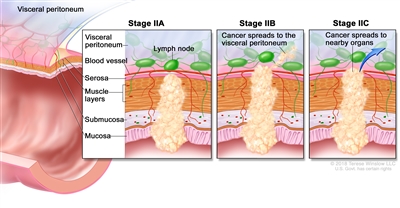
Stage II rectal cancer. In stage IIA, cancer has spread through the muscle layer of the rectum wall to the serosa. In stage IIB, cancer has spread through the serosa but has not spread to nearby organs. In stage IIC, cancer has spread through the serosa to nearby organs.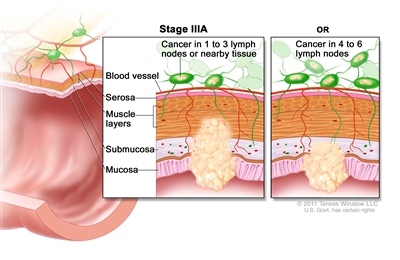
Stage IIIA rectal cancer. Cancer has spread through the mucosa of the rectum wall to the submucosa and may have spread to the muscle layer, and has spread to one to three nearby lymph nodes or tissues near the lymph nodes. OR, cancer has spread through the mucosa to the submucosa and four to six nearby lymph nodes.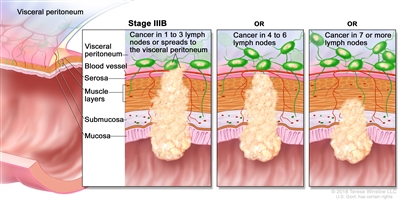
Stage IIIB rectal cancer. Cancer has spread through the muscle layer of the rectum wall to the serosa or has spread through the serosa but not to nearby organs; cancer has spread to one to three nearby lymph nodes or to tissues near the lymph nodes. OR, cancer has spread to the muscle layer or to the serosa, and to four to six nearby lymph nodes. OR, cancer has spread through the mucosa to the submucosa and may have spread to the muscle layer; cancer has spread to seven or more nearby lymph nodes.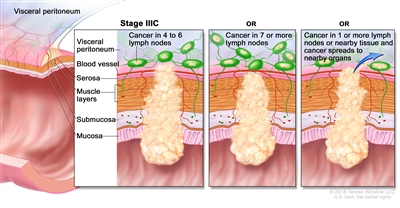
Stage IIIC rectal cancer. Cancer has spread through the serosa of the rectum wall but not to nearby organs; cancer has spread to four to six nearby lymph nodes. OR, cancer has spread through the muscle layer to the serosa or has spread through the serosa but not to nearby organs; cancer has spread to seven or more nearby lymph nodes. OR, cancer has spread through the serosa to nearby organs and to one or more nearby lymph nodes or to tissues near the lymph nodes.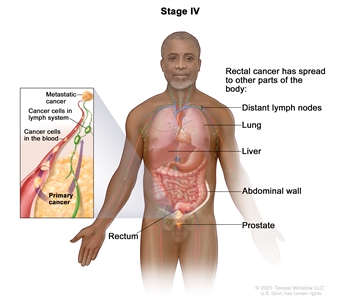
Stage IV rectal cancer. The cancer has spread through the blood and lymph nodes to other parts of the body, such as the lung, liver, abdominal wall, or prostate.Treatment Option Overview
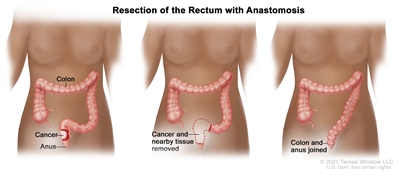
Resection of the rectum with anastomosis. The rectum and part of the colon are removed, and then the colon and anus are joined. orTreatment of Stage 0 (carcinoma in situ)
Treatment of Stage I Rectal Cancer
Treatment of Stages II and III Rectal Cancer
Treatment of Stage IV and Recurrent Rectal Cancer
To Learn More About Rectal Cancer
About This PDQ Summary
Our Health Library information does not replace the advice of a doctor. Please be advised that this information is made available to assist our patients to learn more about their health. Our providers may not see and/or treat all topics found herein.Rectal Cancer Treatment (PDQ®): Treatment - Patient Information [NCI]



